Compatibility of metals

Corrosion is an electrochemical process.
By Rod Newbold, chairman MRM Technical Committee. Article courtesy of Roof Link
In the case of steel, differences in electrical potential exist on the surface, and microscopic cathodic (-) and anodic (+) cells are formed. The positively charged ions in the anodic surface react with negatively charged hydroxyl ions in water to form iron oxide, or rust. Over time polarisation changes cause anodic areas to become cathodic, and vice versa, so that eventually the rate of corrosion is relatively uniform over the surface.
This reaction can only take place in the presence of an electrolyte, e.g. water. The presence of salt in the water increases its conductivity and therefore speeds up the reaction. It also alters its PH level, alkaline and acidic environments will also alter the rate of oxidisation.
Generally, aluminium dominant coatings do better in an acidic environment, but zinc dominant coatings perform better in an alkaline environment. That iswhy zinc dominant systems are preferred in buildings such as enclosed animal shelters and fertiliser sheds, where alkalinity may be high.
Metals are ranked by their electro potential, less noble metals will act as an anode when in contact with more noble metals, and therefore will corrode inpreference to the less noble metal. In the case of zinc coated steel, while the zinc will corrode in preference to the steel, it will generally do so at a slower rate than that shown by unprotected steel, and can therefore give long lasting durability to the substrate.
This table is only an indicator, as some metals react with the atmosphere to form passive surfaces which renders them less active. However it is a helpful guide. Water can flow from a metal higher on the list to one below and metals close to each other on the table are more likely to be compatible in contact. (Caution, the table would suggest that some metals such as lead, monel, stainless and graphite should be more compatible with aluminium dominant coatings than they are with zinc, but that is not the case).
Inert catchment
There is a misunderstanding that Zincalume is incompatible with galvanised surfaces. Zincalume is an alloy of aluminium and zinc, which are adjacent to each other on the galvanic table. They are, as can be expected, compatible in contact. However the aluminium content in Zincalume reacts with the atmosphere to form a stable protective layer of aluminium oxide, which is inert.
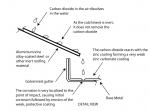
Run-off from inert surfaces such as glazed tiles, aluminium and aluminium/zinc dominant metallic coatings, fibreglass, pre-coated metals, glass or any painted surface can cause corrosion of unpainted galvanised steel and other zinc dominant metallic coatings. This is known as ‘drip-spot corrosion’ or ‘inert catchment corrosion’.
Water sitting on a surface will absorb carbon dioxide forming carbonic acid, which is reactive with zinc. On a galvanised surface, the carbonic acid will react with the zinc and become neutral. On an inert surface discharging into an unprotected zinc surface, the carbonic acid is not neutralised and reaction will be concentrated on the drip points of the inert surface onto the zinc surface.
As the formation of carbonic acid takes time to occur, inert catchment corrosion is normally seen at specific drip points of dew off a roof, rather than below rain-washed painted walls and windows.
Compatibility with other materials
The compatibility chart in the Code of Practice has been updated.
- We are recommending that wet contact between metal and butyl rubber is avoided. Tests are currently under way with several membranes to test their relative compatibility.
- We have added Cedar as this is known to be corrosive.
- We have replaced CCA treated timber with Wet Timber. Tests done by BRANZ of fasteners into treated and untreated timber show little variance in screw corrosion rates embedded into CCA treated and untreated samples. Time of wetness was the critical element
- We have been more cautious about contact between painted aluminium and other surfaces.This is because with painted aluminium, the surface area of the anode can be very small, creating pinhole corrosion.